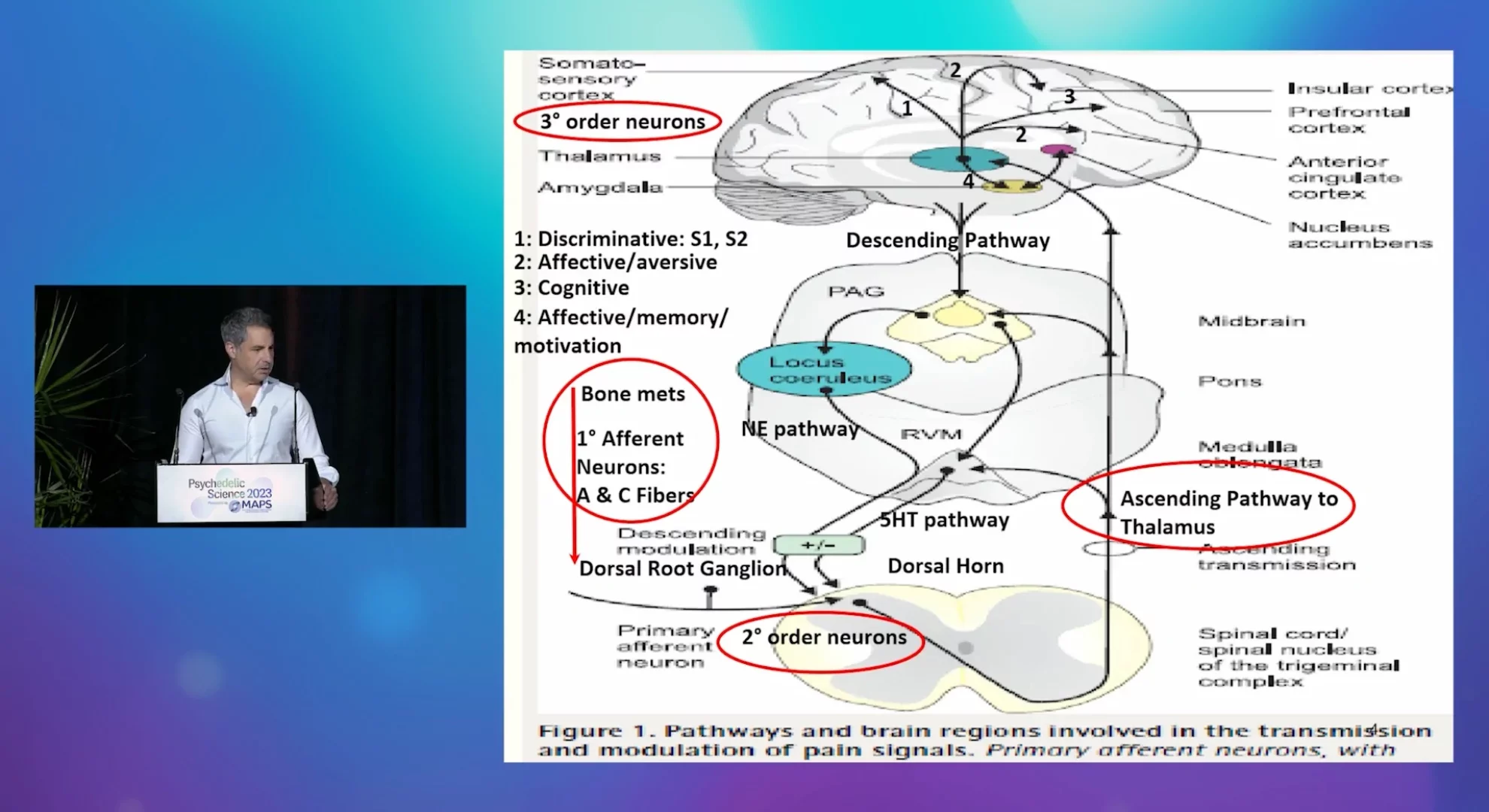
A randomized controlled trial of LSD-assisted psychotherapy to treat metastatic cancer induced bone pain
Metastatic breast cancer-induced bone pain (CIBP) is one of the most common types of pain in cancer, affecting up to 75% of patients with advanced stage breast cancer. CIBP is marked by severe chronic and breakthrough pain, decreased survival and quality-of-life (QoL), as well as substantial co-morbidity of psychiatric and existential distress. There are significant gaps in the pharmacologic treatment algorithm of CIBP including: limited anti-pain efficacy, lack of improvements in QoL, and the potential for serious adverse events (SAEs). In addition, none of the current treatments affect the significant psychiatric co-morbidity associated with CIBP. Despite opioids having a central role in treating CIBP, they carry a considerable side effect burden of somnolence, cognitive impairment, opioid-induced hyperalgesia, addiction, respiratory suppression and risk of overdose fatalities. Thus, there is a critical need for more effective FDA-approved pharmacologic interventions for patients with CIBP. Ideal interventions should be non-addictive, have rapid and sustained analgesic effects, a safe side effect profile, efficacy in treating psychiatric and existential distress, and the potential for opioid sparing.
Lysergic acid diethylamide (LSD) is an ergoline psychedelic with pleiotropic pharmacologic properties including serotonergic receptor agonism producing the well-known subjective effects at the serotonin (5‑HT) 2A receptor, as well as dopaminergic, adrenergic, and glutamatergic agonistic effects. LSD also exhibits nanomolar affinity for many other serotonin receptors, some of which (i.e. 5-HT1B, 1D, 2B, and 7) have been validated as pain targets for which high-affinity receptor agonists are active in rodent pain models. In humans, LSD has been extensively studied (>40,000 participants), with an established favorable safety profile with no significant medical toxicity, lack of addictive liability, and even preliminary efficacy to treat addictive disorders, especially alcohol use disorder, as well as pain and psychiatric and existential distress in end-of-life cancer. From the 1960s -1970s, open-label studies of single to multiple dose LSD-assisted treatment in terminal cancer-related pain syndromes, often with concomitant opioids, reported safety of drug administration, and suggested therapeutic effects of rapidly-acting analgesia that endured for weeks, improvements in psychiatric (depression, anxiety, insomnia) and existential distress (death anxiety), as well as reductions in opioid use. In a comparative efficacy trial of single-dose LSD vs single-dose opioids (meperidine; hydromorphone) in terminal cancer pain, LSD was more effective at pain relief days to weeks after drug administration, compared to the opioids. However, no blinded, placebo-controlled modern trials have been conducted to rigorously determine the potential utility of LSD-assisted therapy for the treatment of CIBP due to bone metastases.
The proposed study is a randomized, double-blind, active-placebo-controlled, parallel group trial of two medication sessions of oral administration of LSD (100 µg; 200 µg µg; 4 weeks apart) combined with manualized psychotherapy sessions to treat metastatic breast CIBP. The primary endpoint is change of pain from baseline to week 12, and secondary and endpoints include opioid sparing, QoL, functional disability, anxiety, depression, sleep, and existential distress. This study may serve as proof-of-concept for LSD therapy as a potentially safe and efficacious treatment for CIBP and other types of cancer pain as well as for refractory chronic neuropathic and musculoskeletal pain.
Share: A randomized controlled trial of LSD-assisted psychotherapy to treat metastatic cancer induced bone pain
Facebook
Twitter
LinkedIn
Email




